What is cold chain validation?
Cold chain validation is the GxP process of proving that every temperature-controlled environment your product passes through performs within defined limits and produces audit-ready evidence. Most validation programs still run on a periodic cycle – a study, a report, a multi-year wait until the next one.
That model is increasingly hard to defend. FY2025 set a 20-year record for FDA warning letters, with environmental monitoring deficiencies cited in more than 15% of them. This page covers what cold chain validation includes under EU GDP, WHO TRS 961 Annex 9, USP <1079>, and FDA 21 CFR Part 211, where the periodic model is breaking down, and how continuous validation works in practice.
Get a template to use as basis for your cold chain validations based on GDP, WHO, USP, and FDA requirements.

What does cold chain validation include?
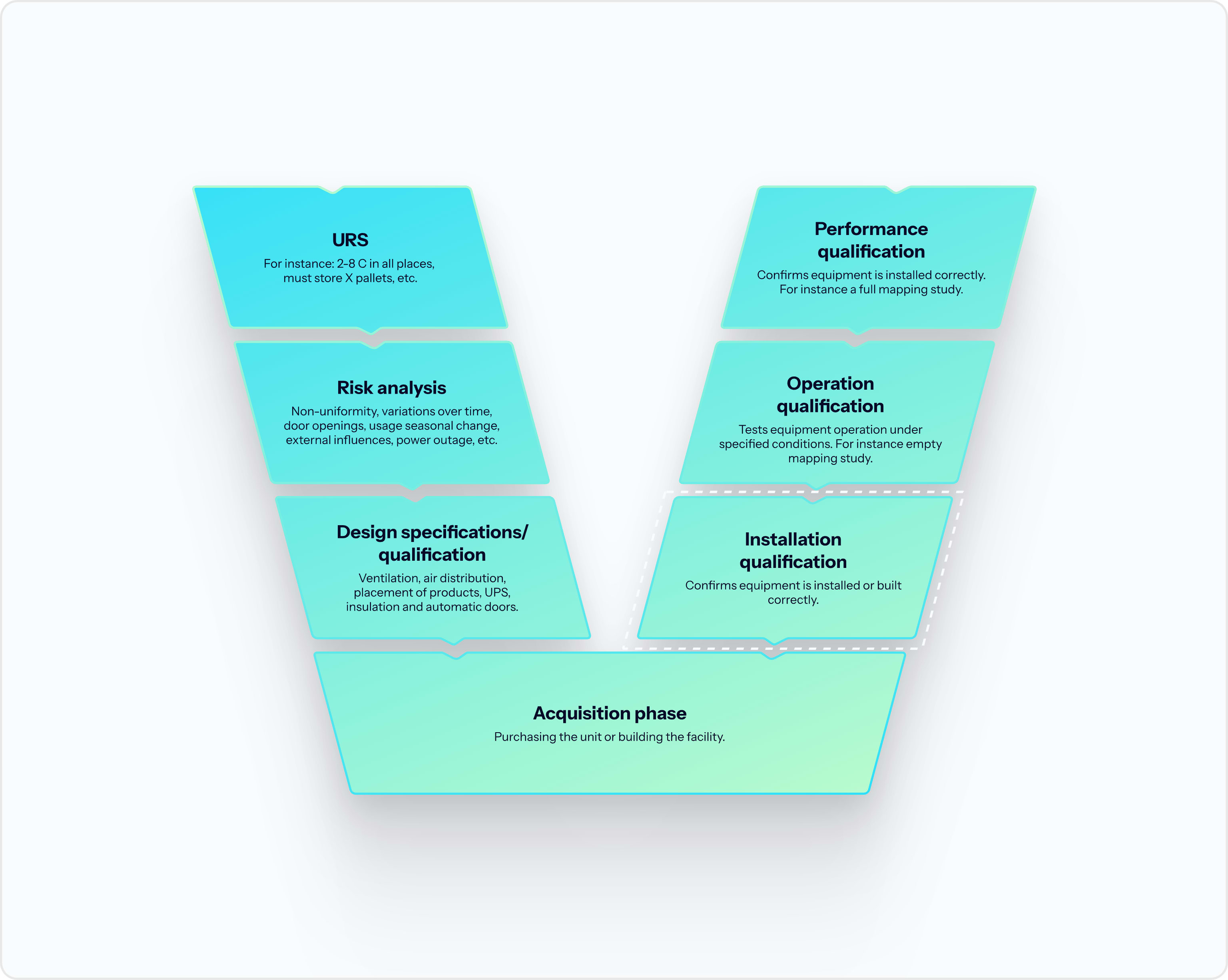
Cold chain validation is the umbrella process. A complete validation file is built from four activities: qualification of individual equipment through DQ, IQ, OQ, and PQ; mapping to identify worst-case zones inside a facility or unit; monitoring to maintain the validated state with ongoing data; and calibration to keep the monitoring evidence traceable under ISO 17025.
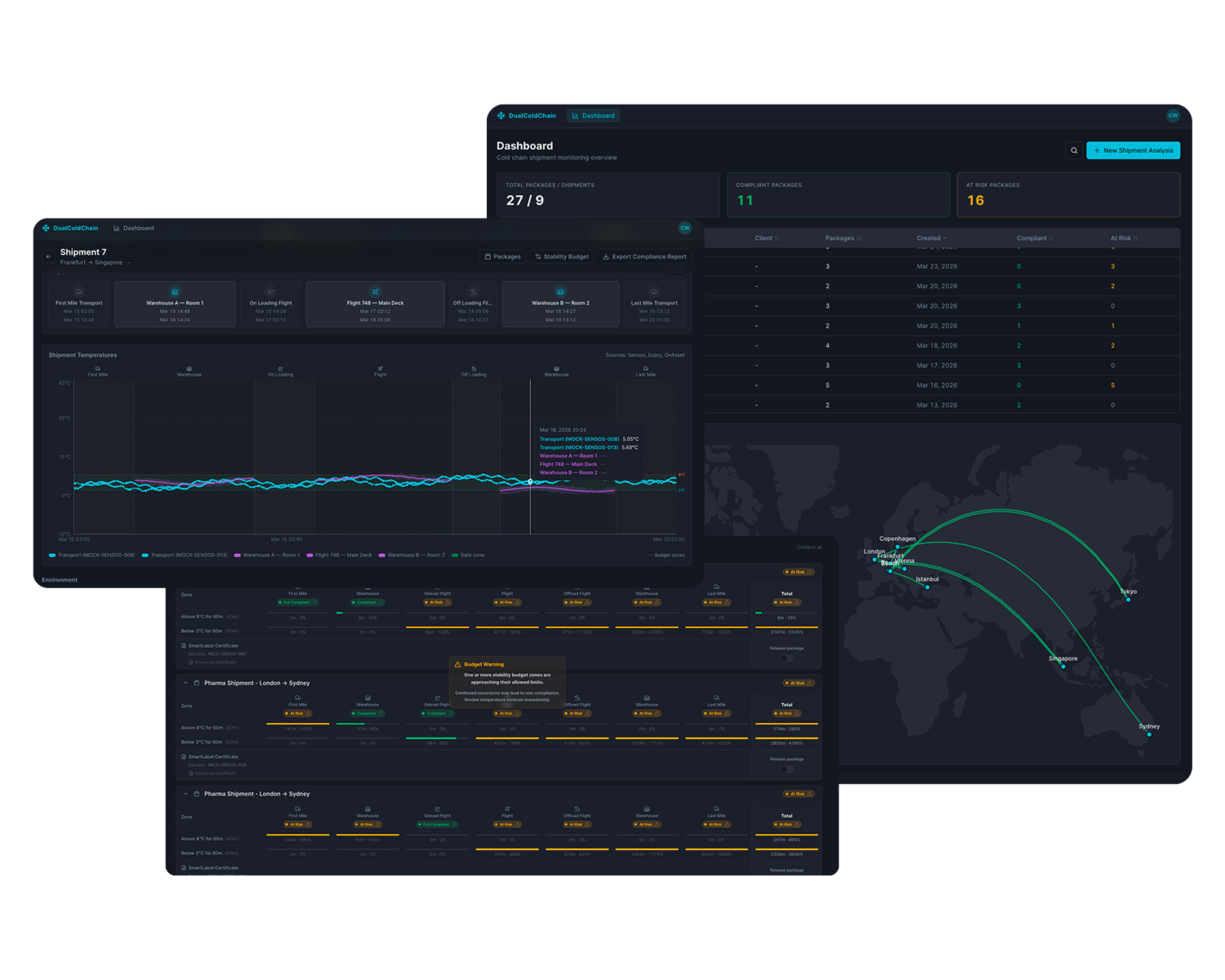
The Eupry Compliance Platform consolidates all four on the same hardware, in one evidence trail.
*Also read: What is thermal validation?

Periodic validation cannot keep up
The protocol-driven model was built for a smaller, slower industry. It produces a snapshot: a study, a report, a sign-off, then a multi-year wait until the next cycle. In current GxP environments, four structural problems are showing up repeatedly in inspection findings.
One parameter is no longer enough
FDA inspectors are citing facilities for monitoring temperature when the URS specifies humidity, CO₂, or differential pressure, too. Single-parameter monitoring against a multi-parameter requirement is one of the most common warning-letter triggers.
Multi-site scheduling chaos
Validation studies stack up on growing equipment estates. Vendors compete for calendar slots. The file looks complete on paper, while the underlying evidence drifts further from operating reality.
The control point is wrong
Per-shipment monitoring assumes facilities are unvalidated, so every package needs its own device. Costs scale linearly with volume forever, and the data lands after products are already compromised.
Handoffs go unowned
Loading docks, tarmacs, and receiving bays sit between validation files. When environmental monitoring deficiencies show up in warning letters, the citation is often at exactly these transition points.
Validate continuously, not periodically
Continuous validation replaces the periodic study with a permanent record. Sensors stay in place, data flows continuously, and the validation file is reconstructed from live evidence rather than scheduled studies. The framework is risk-based, GxP-aligned, and supported by published Validation 4.0 thinking.
One vendor, all you need
Cold chain monitoring, calibration, and validation in one platform. No more coordinating suppliers or systems.
Track all parameters
Temperature, humidity, CO₂, and differential pressure on one dashboard. The parameters auditors expect documented, in one evidence trail.
Fully automated
Generate audit reports in 3 clicks. No manual downloads, no spreadsheets, no piecing together data.
Live monitoring
Real-time monitoring with stability budget tracking lets you prevent losses instead of documenting them.
Sensors placed once
Documented risk assessment identifies worst-case zones. Sensors stay in place for ongoing monitoring and mapping.
Calibration on the wall
Patented sensor-tip technology keeps loggers traceable under ISO 17025 without removing them from operation.
What are the differences between periodic and continuous cold chain validation?
Most QA teams know the periodic model. The question is what changes when validation runs continuously. Five differences show up consistently in audit prep and multi-site oversight.
Evidence
- Periodic: snapshot from a study, every 1–3 years
- Continuous: live record, always current
Re-validation trigger
- Periodic: calendar
- Continuous: change
Multi-site oversight
- Periodic: spreadsheet of due dates
- Continuous: Compliance Status Dashboard
Coverage
- Periodic: storage units in isolation
- Continuous: storage, transit, and handoffs
Audit prep
- Periodic: weeks
- Continuous: three clicks


Download a cold chain validation protocol
Get a a working template for documenting validation across storage, transit, and receipt under EU GDP, WHO, USP, and FDA requirements.
How does Dual Compliance for cold chain validation work?
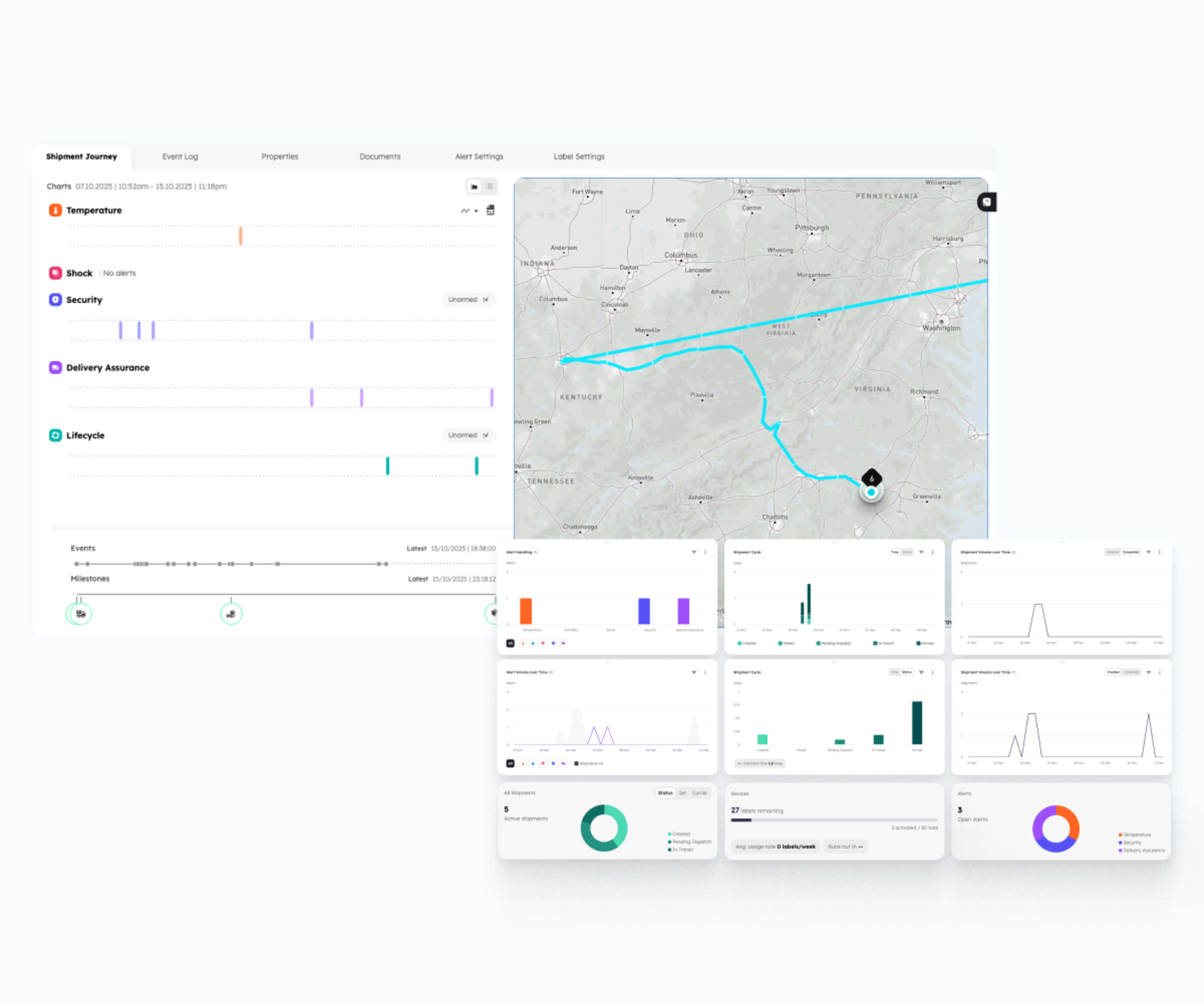
Continuous facility monitoring covers everything passing through validated environments. Some shipments still warrant individual attention – high-value biologics, new routes, or specific regulatory requirements. The Dual Compliance Platform combines both in one record.
- Layer one – environmental monitoring. Wireless data loggers in warehouses, aircraft cargo holds, and trucks. A validated environmental record for every product that passes through, whether or not it carries its own tracking device.
- . Layer two – Smart Label shipment tracking. Strategic, not universal. Temperature, humidity, GPS, shock, and tamper events for the shipments that warrant individual monitoring.
Both layers correlate automatically across every handoff point. Stability budgets are tracked live across the full journey. A product with a 60-minute budget outside +2°C to +8°C might use 5 minutes in a warehouse and 35 minutes in an aircraft cargo hold – 40 of 60 minutes used, still compliant. Without live tracking across every leg, that cumulative total is invisible until after delivery.
Most organizations begin with dual verification – running both layers in parallel during an initial validation period to build statistical evidence – and transition to dual compliance as the operating model once correlation is proven.
Also see: What is dual compliance?

How does cold chain validation cover storage, transit, and receipt?
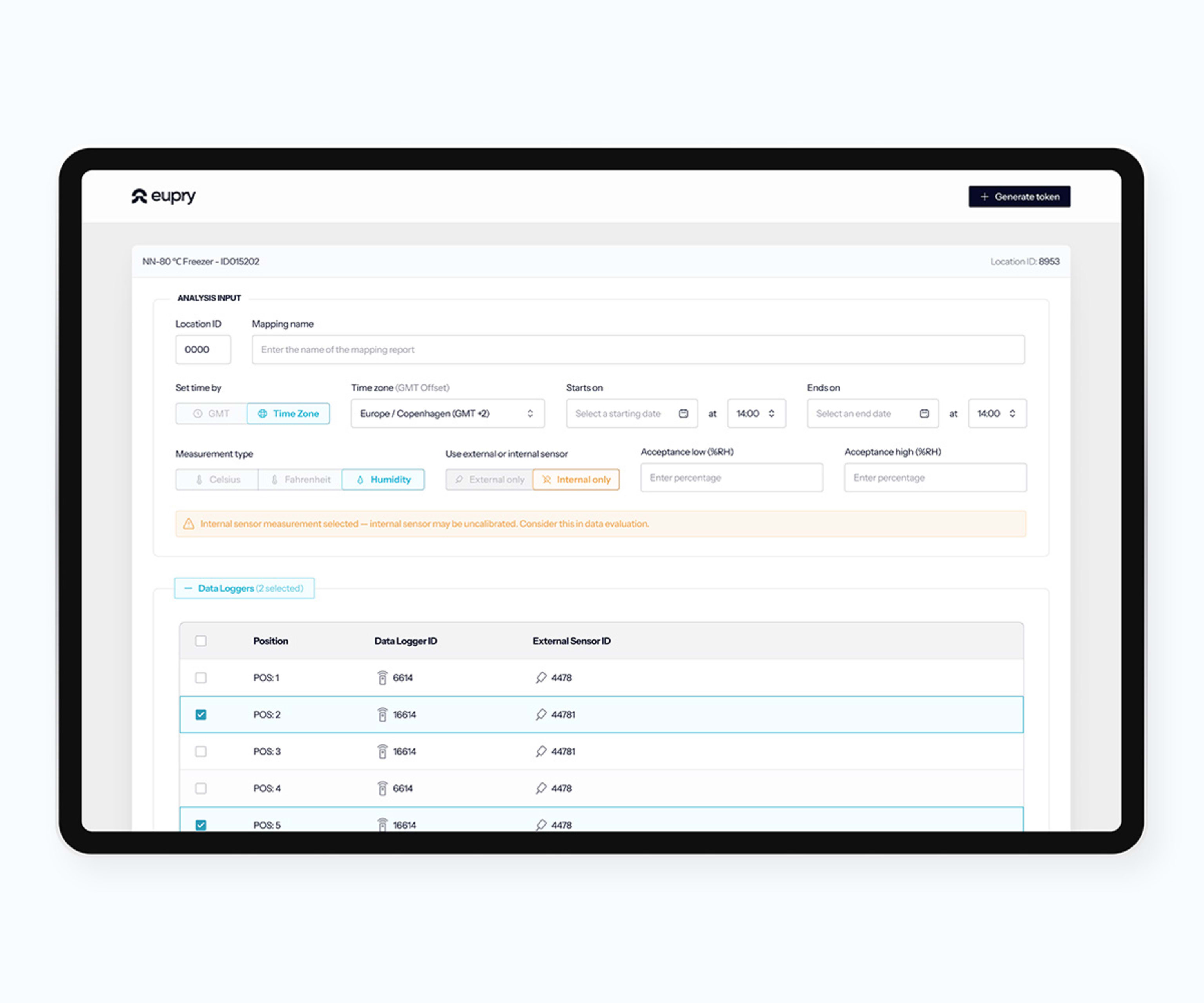
- Storage: wireless data loggers and EasyMap cover cold rooms, freezers, refrigerators, stability chambers, incubators, and ULT freezers.
- Vehicle transit: trucks and last-mile vehicles are treated as continuously monitored assets, qualified the same way a warehouse is.
- Air freight: the world's only FAA-approved loggers for permanent installation in aircraft cargo holds. Continuous mapping data within 2 minutes of landing.
- Receipt: the Compliance Status Dashboard surfaces conditions at the receiving site alongside storage and transit, so handoffs are visible rather than implied.
Also read: Aircraft CMM

What auditors look for in your cold chain validation evidence
Built for GDP, GMP, and FDA scrutiny
Inspectors grade validation files on a small number of structural questions. A file that answers these cleanly will hold up under inspection.
- Risk-based justification: sensor placement, monitoring frequency, and revalidation triggers tied to a documented risk assessment. Eupry's mapping methodology is risk-based by default.
- Every parameter in the URS: monitoring evidence covering each parameter the product specification requires. Multi-parameter coverage is one of the most common audit focus areas.
- Data integrity under 21 CFR Part 11 and Annex 11: audit trails, electronic signatures, and time-stamped records throughout. Eupry is FDA 21 CFR Part 11 compliant and certified to ISO 27001.
- Coverage across handoffs: monitoring records for loading docks, transit windows, and receipt bays alongside the facilities they connect.
- A current file: continuous monitoring keeps validation evidence live by design.

Also see: Audit checklist for GDP | GxP compliance

Continuous cold chain validation with Eupry
One platform for every parameter, every stage, audit-ready by design.
- Mapping and monitoring: continuous, on the same hardware, across temperature, humidity, CO₂, and differential pressure
- Calibration: patented on-the-wall sensor-tip technology, traceable under ISO 17025
- Transit: validated environmental records for warehouses, aircraft cargo holds, and trucks
- Audit reports: three clicks under FDA 21 CFR Part 11 and EU GMP Annex 11

From 5, 10, 40+ vendors to 1
Most pharma operations juggle separate vendors for monitoring, calibration, mapping, and validation services.
With Eupry, you get one integrated solution.
- One vendor relationship, contract, and support team
- One compliance framework across all sites and parameters
- Predictable, reduced compliance budget at scale

Trusted by 1000+ companies worldwide

Download a cold chain compliance catalog
Get an instant overview of how the cold chain monitoring system and validation service works in the product catalog.
Compatible products & add-ons
Cold chain validation FAQ
Get started with cold chain monitoring
Keep your product safe at every step of the journey.